We've quantified the ethical and scientific costs of inadequate antibody validation — and developed an evidence-based, consensus-driven strategy to fix it. Here's what we found, what needs to happen, and the tools to make it easier.
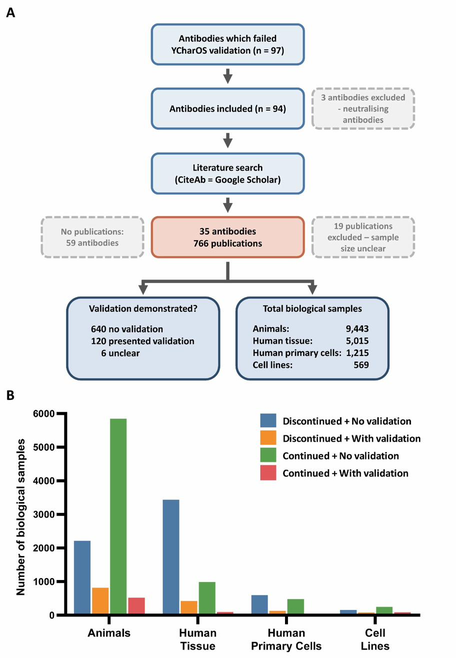
Independent characterisation by YCharOS using standardised knockout-control protocols demonstrated that over 50% of 614 commercial antibodies failed in at least one application. The eLife paper by Kahn, Virk, Laflamme et al. (2024) proposed sector-specific actions. An NC3Rs stakeholder meeting (February 2024, 56 participants) brought together manufacturers, publishers, funders, and researchers to identify priority interventions. But the downstream ethical consequences had never been quantified.
Using focus groups (n=12), a national survey (n=107), and systematic analysis of 785 publications, we quantified for the first time how many animal and human tissue samples are consumed in experiments using antibodies with demonstrated poor performance. We also mapped the behavioural drivers using the COM-B framework — revealing that antibody selection is dominated by social proof and habit rather than systematic evaluation of performance data.
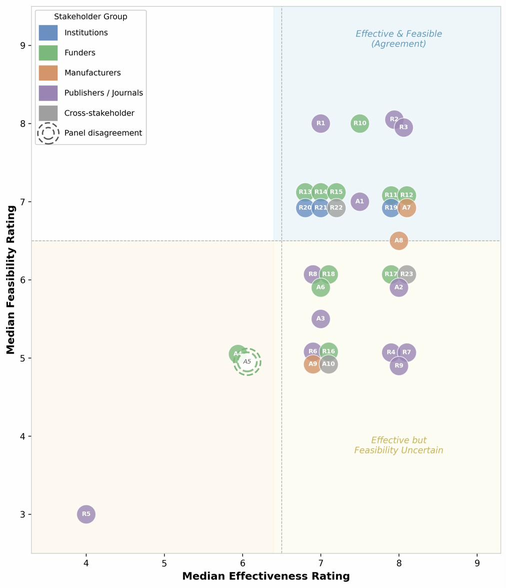
Read the preprint →A modified Delphi study with 32 international experts — representing researchers, publishers, funders, manufacturers, and institutional leaders — produced 15 consensus recommendations rated as both effective and feasible for implementation by 2030. The panel endorsed a staged approach: enablement first (training, resources, infrastructure), then expectation (requirements, standards), then mandate (enforcement). Each stakeholder group has a defined role.
Read the preprint →The OGA database, Academy, and Antibody Champions programme provide practical infrastructure to support implementation. We're bringing stakeholders together — including at the UKRN Annual Conference in July 2026 — to turn recommendations into coordinated action.
Mixed-methods study quantifying the ethical costs of poor antibody validation for the first time — including avoidable use of animal and human biological materials.
Modified Delphi study producing actionable, stakeholder-specific recommendations for improving antibody validation, selection, and reporting practices.
Training, research integrity frameworks, and local champions
Validation budgets, grant requirements, reporting standards, benchmarking
Antibody reporting packages, validation standards, editorial capacity
Product identification (RRIDs at source), validation data, recombinant transition
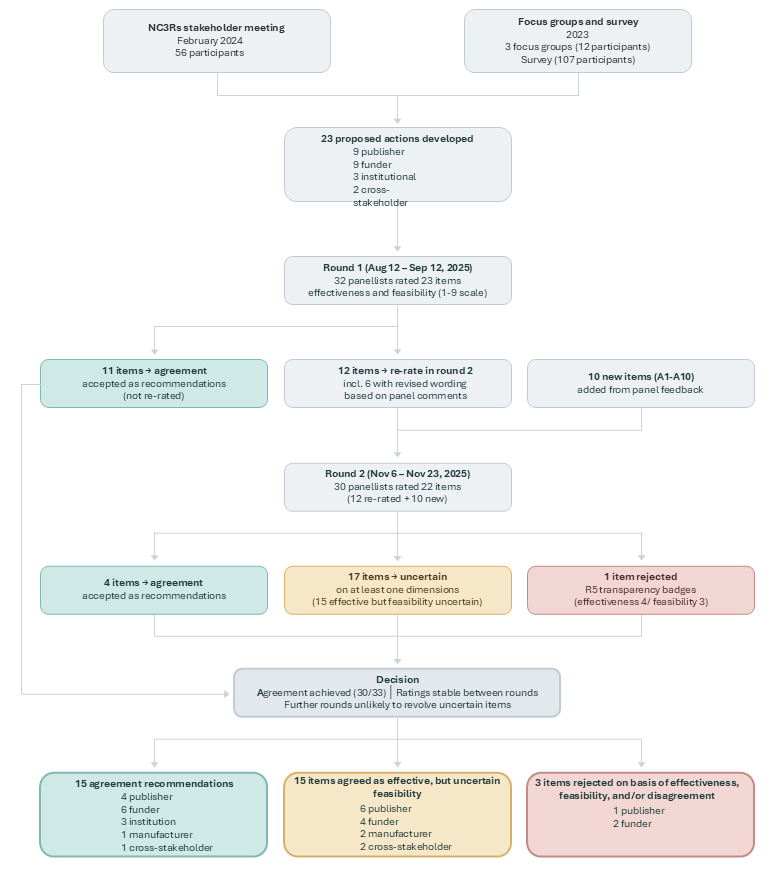
The Delphi panel included senior representatives from organisations including Abcam, Cell Signaling Technology, Proteintech, GeneTex, AstraZeneca, the Institute for Protein Innovation, Addgene, YCharOS/SGC, eLife, Nature Protocols, F1000, Wiley, The Company of Biologists, NC3Rs, the Michael J. Fox Foundation, the MND Association, CHDI Foundation, CiteAb, SciCrunch, and leading research institutions. This work also builds on the independent characterisation data generated by the YCharOS consortium.
View all OGA partners →Searchable characterisation data with knockout controls across Western Blot, IP, ICC/IF, and flow cytometry. Find out which antibodies actually work for your target — and which don't.
Structured e-learning covering antibody selection, validation strategies, database use, and data interpretation. Free, self-paced, with assessment and certification.
A joint OGA/NC3Rs programme recruiting early career researchers to drive culture change in antibody validation at their own institutions. 12-month programme with professional training and expert mentorship.
We have a 90-minute panel session — "Working together to improve biomedical research: the OnlyGoodAntibodies experience" — at the UK Reproducibility Network Annual Conference, 8–9 July 2026.
Conference details →We're forming working groups to develop practical implementation guidance — including model curricula, institutional policy templates, and coordination mechanisms. Share your feedback or express interest in joining.
Get in touch →